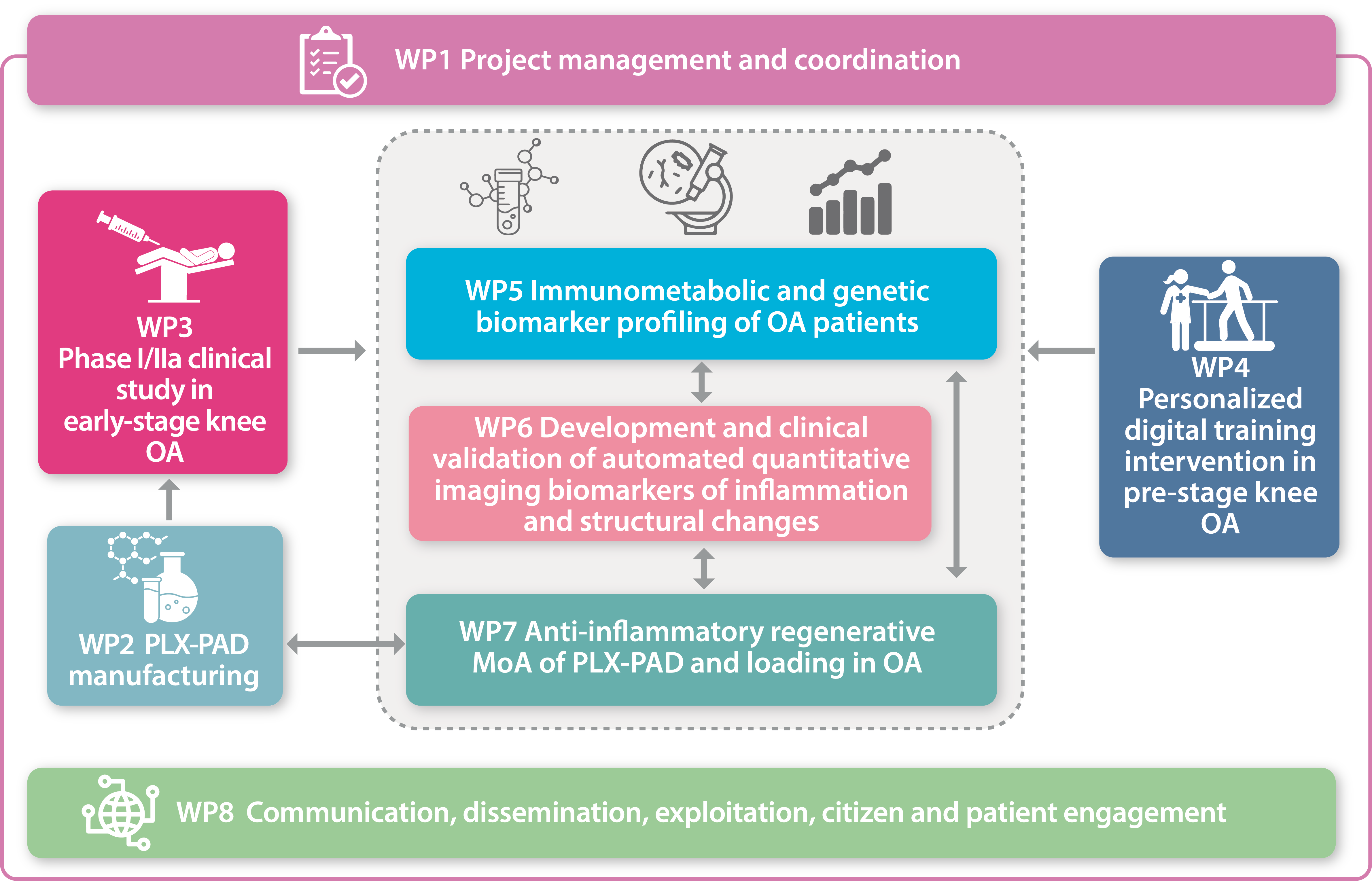
The Challenge

Osteoarthritis (OA) is a chronic progressive joint disorder, characterized by inflammation causing pain, stiffness, swelling and gradual loss of joint function. Within PROTO we have joined forces to address the key feature of OA progression – inflammation. We intend to do this in a two-pronged approach addressing different early inflammatory disease stages.
OA affects over 16% of the global adult population, and it is the most common and rapidly growing form of arthritis, leading to reduced mobility and chronic disability and is associated cardiovascular and metabolic co-morbidities. OA is mostly triggered by micro- and macro-injuries to affected joints, where repair processes initiate proinflammatory immune cascades that eventually lead to progressive joint destruction. This vicious cycle results in cartilage degradation, subchondral bone remodelling and low-grade joint inflammation. To date there are no disease modifying treatments that can prevent or slow down OA.
Within PROTO we have joined forces to address the key feature of OA progression – inflammation.
We intend to do this in a two-pronged approach addressing different early inflammatory disease stages.
- We will use an innovative anti-inflammatory allogeneic placental (PLX-PAD) cell therapy to treat patients with early-stage knee OA as a direct approach to reduce inflammation and degeneration.
- We will train patients following anterior cruciate ligament reconstruction (ACLR) 12-24 months after surgery, to restore normal biomechanics and alleviate mechanically induced inflammation within the joint. This group will serve as a pre-stage knee OA group, as in previous work we have shown that pathological gait patterns in persons after ACLR or with knee OA are associated with progressive cartilage loss and persistent inflammation.
Ambitions
PROTO aims to halt and partially reverse the structural and functional changes caused by inflammatory processes in OA Our ambitious goal is to introduce:
- a highly innovative anti-inflammatory local placental derived cell therapy in early-stage OA patients and
- a personalized sensor-based training intervention intended to prevent inflammation and OA onset during a crucially important ‘window of opportunity’ by correcting pathological movement patterns in pre-stage OA patients.

Central to both studies will be the thorough characterization and analysis of the influence of the interventions on the local and systemic inflammatory status of the patients. Cutting edge biomarker studies, which have been fed by mechanistic knowledge gained from consortia data will accompany both intervention studies. Both studies are early proof-of-concept studies with a sound preclinical scientific basis and aim to revolutionize OA prevention and therapy.
Objectives

Within the funding period PROTO aims to create a reliable, sustainable and evidence-based concept for tackling inflammation in pre- and early-stage OA, which can be used in future therapeutic interventions to prevent OA development and progression.
Within the funding period PROTO aims to:
- To investigate the safety and efficacy of a local anti-inflammatory PLX-PAD cell therapy to halt and partially revert early-stage knee OA in a randomized, double blind, placebo-controlled phase I/II trial.
- To understand the anti-inflammatory MoA of PLX-PAD in patients with early-stage knee OA, as seen in preclinical and clinical trials for other indications, and to correlate the efficacy of the treatment to the patients´ immune experience using a well standardized immune monitoring approach.
- To define the PLX-PAD immune-mediated effect on joint structure and function through a potentially reshaped anti-inflammatory intraarticular environment using physiologically relevant and biomimetic in vitro and ex vivo MoA studies.
- To prevent health-to-disease transition in pre-stage OA patients, we will implement a training intervention to correct pathological movement patterns with a sensor- and app-based digital tool.
- We will measure and monitor the influence of a personalized training intervention on the inflammatory status of the knee joint in pre-stage OA patients.
- To create a reliable, sustainable and evidence-based concept for tackling inflammation in pre- and early-stage OA, which can be used in future therapeutic interventions to prevent OA development and progression.
- To achieve increased communication, better engagement, and expanded insights into the perspective of participating patients by the inclusion of a patient portal (BtP) for empowering patients as active participants in the R&D process.